Login (User Login; top right of page) with your web user or complete this form to download.
Modern HMI for SANDMANN Machines & Systèmes SAS (France)

Modern HMI for SANDMANN Machines & Systèmes SAS (France)
.pdf / 293.07 KB
Nedladdning
Highlights:
- Use of project templates: pharma HMI Application Set
- Configurable audit trail
- Decentralized application on a server and virtual client
- Management of recipe versions
- Generation of customized PDF reports
- SQL database management
- Electronic signature for authentication
SANDMANN Machines & Systèmes SAS builds machines for engraving a data matrix code on the cap of pharmaceutical vials using a laser and then reading it for quality control purposes. These machines are generally placed downstream of a filling line (for the marking step) and upstream of an assembly or packaging line (for the reading step).
Each vial is marked with a unique and non-modifiable code containing all the information required to identify it, including production site, batch number, and the unique incremental number of the vial in the batch.
In 2021, SANDMANN Machines & Systèmes SAS received a special request from a major international client that specializes in developing products and solutions for healthcare and life sciences. It wanted to equip several production sites with machines to reliably and precisely identify vials containing pharmaceutical products. SANDMANN Machines & Systèmes SAS was an ideal partner for this project because of its longstanding experience in the field.
Preventing mix-ups and ensuring a strong track-and-trace system are critical in a pharmaceutical setting. Some products are shipped all over the world before being labeled. Tamper-proof codes guarantee a robust supply chain in the name of patient safety.
In addition, regulatory requirements demand enhanced traceability in order to support logistical distribution and prevent falsification.
The first step in manufacturing the new machines involved compiling a design brief and conducting the factory acceptance test (FAT). These tests were performed at the SANDMANN Machines & Systèmes SAS factory, making it possible to verify the smooth operation of the system and its components. They assessed whether the equipment and its performance satisfied the purchase specifications and analyzed the equipment’s technical documentation to check the compliance of installed components.
During the FAT phase, the client’s expectations weren’t entirely fulfilled in terms of integrating the system with the existing infrastructure or with regard to proposed functions, such as the audit trail, recipe management, and user administration.

The engineers in charge of the project at SANDMANN Machines & Systèmes SAS were conscious of the fact that using the existing software, it would take time to develop. This would potentially increase the costs of manufacturing the machine and delay delivery.
Therefore, they defined new requirements and conducted research to find a suitable alternative solution and evaluated the various alternatives available on the market. For this project, they needed a solution that they could integrate easily in their equipment landscape. It would need to be highly intuitive for users and ensure the prompt delivery of the machine.
Their research led to them proposing a reliable and innovative human-machine interface (HMI) : the zenon software platform from COPA-DATA.
Meeting pharmaceutical requirements
The zenon software platform won over SANDMANN Machines & Systèmes SAS on the basis of its extensive native functions and the ease with which it could be integrated with the existing environment.
A plan was subsequently put in place with the client, consisting of training for the zenon interface, an introduction to the zenon pharmaceutical Application Set, and a number of expertise days during which COPA-DATA walked the engineers at SANDMANN Machines & Systèmes SAS through configuring the advanced functions.
COPA-DATA’s Application Set provides preconfigured projects that meet the applicable standards and offer templates that serve as a basis for developing new projects.
“The ergonomic design of zenon was a particular strong point for us because it allowed us to save a considerable amount of time in developing the project. The native functions offered by zenon speed up development thanks to their straightforward configuration, while most other solutions require lengthy development,” says Ludovic Malherbe, head of the automation project at SANDMANN Machines & Systèmes SAS.
With zenon, the machine user now has a complete overview of current operations, as well as the ability to ensure the safety and quality of products.
This new machine incorporates a wide range of native zenon functions, which are preconfigured in the Application Set to deliver a solution that meets the requirements of the pharmaceutical industry:
- User authentication integrated in the client’s IT system (Active Directory) and an electronic signature procedure for changing critical parameters (double signature)
- Management of product conformities and non-conformities (list of non-compliant products for checking purposes)
- Recipe management (managing different vial sizes, entering new recipes on a graphical display, managing recipe versions)
- Implementation of an audit trail for ensuring the traceability of critical parameters
- Advanced reports: batch reports and production reports available as PDFs
- Network technology (decentralized application on a server and virtual client)
- Opportunities for further analytics via export to an SQL database
A project turned success story
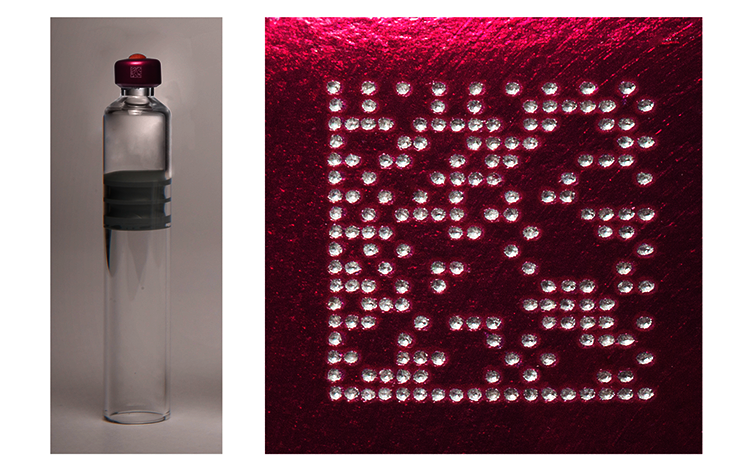
The client considered the ergonomic design of zenon to be a real asset.
zenon offers preconfigured project templates that simplify project development, management and maintenance. In just a few clicks, SANDMANN Machines & Systèmes SAS was able to configure new projects to cater to the requirements of the pharmaceutical market and its client’s expectations. zenon’s intuitive HMI provides a clear overview that is easy to navigate and to find all the necessary information.
“When the client tested zenon, they knew they had made the right choice and immediately praised us for selecting this solution,” reports Ludovic Malherbe.
The installation and configuration of the machine were thereby made easier, allowing SANDMANN Machines & Systèmes SAS to meet its commitments in terms of the delivery timeframe and the functions offered.
The new unique ID laser marking machine now manages hundreds of thousands of items a day.
Back